
Tumor Cells’ Inner Workings Predict Cancer Progression
Molecular markers help reveal nature of chronic lymphocytic leukemia – slow or fast
By:
- Scott LaFee
Published Date
By:
- Scott LaFee
Share This:
Article Content
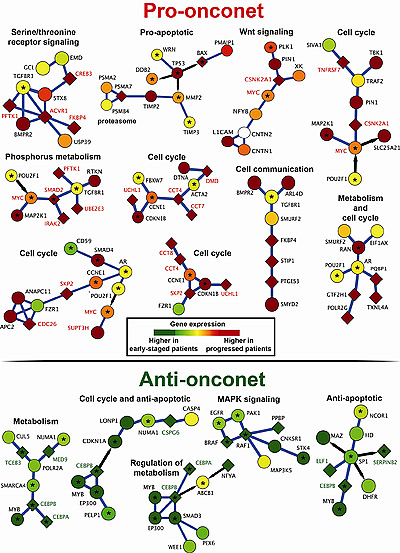
The schematic above depicts some of the subnetworks of interacting proteins that either promote or slow the progression of CLL.
Using a new assay method to study tumor cells, researchers at the University of California, San Diego School of Medicine and UC San Diego Moores Cancer Center have found evidence of clonal evolution in chronic lymphocytic leukemia (CLL). The assay method distinguishes features of leukemia cells that indicate whether the disease will be aggressive or slow-moving, a key factor in when and how patients are treated.
The findings are published in the July 26, 2012 First Edition online issue of Blood.
The progression of CLL is highly variable, dependent upon the rate and effects of accumulating monoclonal B cells in the blood, marrow, and lymphoid tissues. Some patients are symptom-free for years and do not require treatment, which involves the use of drugs that can cause significant side effects and are not curative. In other patients, however, CLL is relatively aggressive and demands therapeutic intervention soon after diagnosis.
“Our study shows that there may not be a sharp dividing line between the more aggressive and less aggressive forms of CLL,” said Thomas J. Kipps, MD, PhD, Evelyn and Edwin Tasch Chair in Cancer Research and senior author of the study. “Instead, it seems that over time the leukemia cells of patients with indolent disease begin to use genes similar to those that are generally used by CLL cells of patients with aggressive disease. In other words, prior to requiring therapy, the patterns of genes expressed by CLL cells appear to converge, regardless of whether or not the patient had aggressive versus indolent disease at diagnosis.”
Existing markers for aggressive or indolent disease are mostly fixed and have declining predictive value the longer the patient is from his or her initial diagnosis. When the blood sample is collected, these markers cannot reliably predict whether a CLL patient will need therapy soon, particularly when the patient has had the diagnosis of CLL for many years.
Kipps and colleagues studied thousands of genes, particularly those that code for proteins, in a group of 130 CLL patients with varying risks of disease progression. They identified 38 prognostic subnetworks of interacting genes and proteins that, at the time of sample collection, indicate the relative the aggressiveness of the disease and predict when the patient will require therapy. They confirmed their work using the method on two other, smaller CLL patient cohorts in Germany and Italy.
The subnetworks offer greater predictive value because they are based not on expression levels of individual genes or proteins, but on how they dynamically interact and change over time, influencing the course of the CLL and patient symptoms.
“In a sense, we looked at families rather than individuals,” said Kipps. “If you find in an interconnected family where most genes or proteins are expressed at higher levels, it becomes more likely that these genes and proteins have functional significance.”
He added that while the subnetworks abound in data, their complexity actually makes them easy to interpret and understand. “It’s like when you look out of a window and see the sky, clouds, trees, people, cars. You’re getting tremendous amounts of information that individually doesn’t tell you much. But when you look at the scene as a whole, you see patterns and networks. This work is similar. We’re taking all of the individual gene expression patterns and making sense of them as a whole. We’re more able to more clearly see how they control and regulate function.”
The findings help define how CLL – and perhaps other cancers – evolve over time, becoming more aggressive and deadly. “It’s as if each tumor has a clock which determines how frequently it may acquire the chance changes that make it behave more aggressively. Although the rates can vary, it appears that tumors march down similar pathways, which converge over time to a point where they become aggressive enough to require therapy.”
The study may alter how scientists think about CLL and how clinicians treat the disease: whether it is better to wait for later stages of the disease when tumor cells are more fragile and easier to kill, or treat early-stage indolent tumor cells aggressively, when they are fewer in number but harder to find and more resistant to therapy.
Co-authors are Han-Yu Chuang and Trey Ideker, Bioinformatics and Systems Biology Program, Department of Bioengineering, Department of Medicine, Moores Cancer Center, all at UCSD; Laura Rassenti, Department of Medicine and Moores Cancer Center, UCSD; Michelle Salcedo, Division of Biological Science, UCSD; Kate Licon, Department of Bioengineering and Department of Medicine, UCSD; Alexander Kohlmann, Roche Molecular Systems, Inc.; Torsten Haferlach, MLL Munchner Leukamielabor GmbH, Germany; and Robin Foa, Division of Hematology, University “La Sapienza,” Italy.
Funding for this research came, in part, from National Science Foundation grant NSF425926, National Institutes of Health grant ES14811, Pfizer and Agilent laboratories. Additional support came from NIH grants for the CLL Research Consortium (P01-CA081534), a Merit Award to Kipps and trainee research award to Chuang from the American Society of Hematology.
About Chronic Lymphocytic Leukemia
CLL is the most common form of leukemia, and most commonly diagnosed in adults after the age of 50. Incidence rates increase with age. The majority of CLL patients are men. Roughly 16,000 Americans are diagnosed with the disease each year; 4,400 die from the disease annually. CLL is currently considered incurable, but typically progresses slowly with no ill effect. Treatment is often postponed until serious symptoms appear that affect quality of life.
Share This:
You May Also Like
Stay in the Know
Keep up with all the latest from UC San Diego. Subscribe to the newsletter today.


