
By:
- Daniel Kane
Published Date
By:
- Daniel Kane
Share This:
Bioengineers Visualize Fat Storage in Fruit Flies

Lingyan Shi
For the first time, researchers have visually monitored, in high resolution, the timing and location of fat storage within the intact cells of fruit flies. The new optical imaging tool from the lab of bioengineering professor Lingyan Shi at the University of California San Diego is already being used to untangle often discussed, yet mysterious, links between diet and things like obesity, diabetes and aging. The work from bioengineers at the UC San Diego Jacobs School of Engineering is published in the journal Aging Cell.
The optical microscopy platform developed by the UC San Diego bioengineers is unique. It allows the researchers to visually track, in high resolution within fat cells, how specific dietary changes affect the way flies turn the energy from their food into fat. The tool also allows the researchers to monitor the reverse process of changing fat back into energy. In addition, the researchers can now visually monitor changes in size in individual fat-storage "containers" within the class of fruit fly cells that is analogous to mammalian fat (adipose) cells.
In the new paper in Aging Cell, the researchers demonstrated the ability to visually track changes in fat (lipid) metabolism in flies after they were put on a wide range of different diets. The diets included calorie-restricted diets, high protein diets, and diets with twice, four-times, and ten-times the sugar of a standard diet.
"With our new optical microscopy system, we can see both where and when fats are being put into storage and taken out of storage," said Shi, the bioengineering professor at UC San Diego who is the corresponding senior author on the new paper. "This is the first imaging technology that can visualize fat metabolism at high resolution in both space and time within individual fat cells. We have demonstrated that we can see both where and when lipid metabolism changes within individual fruit fly fat body cells in response to dietary changes."
"Interest in optimizing the human diet is intense," Shi continued. "People want answers to questions like, 'What are the best diets to slow aging? What are the best diets for losing weight? What are the best diets for extending health span?' I don't yet have answers to these questions, but in my lab, we develop new technologies that are getting us closer to answering some of the big dietary questions out there."
In the new work in Aging Cell, for example, the researchers report a new way to answer questions like:
How much does a specific diet, such as a high-protein diet, or a high-sugar diet, or a calorie-restricted diet, alter a fruit fly's process of turning energy from food into fat? And how much do these same diets affect a fruit fly's process of turning fat back into energy?
"We developed this tool to help us untangle the relationships between diet and phenomena like obesity, diabetes, aging, and longevity," said Shi.
Tracking the size of fat droplets within intact fruit fly cells is one example of what's possible with the new visualization platform.
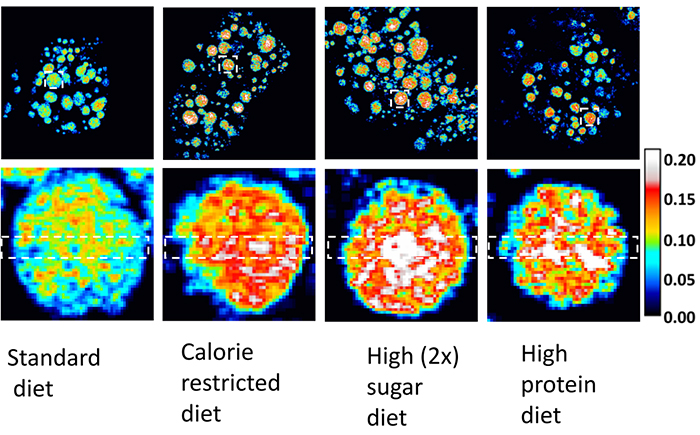
These images were captured by the technology developed in Lingyan Shi's bioengineering lab at UC San Diego. The technology allows researchers to collect high-resolution information, in both time and space, from within individual fat cells of fruit flies. The images in the lower row are zoomed-in portions of the images in the upper row. Together, these images tell a visual story of fat (lipid) metabolism in fruit flies that were fed different diets. The higher the color-coded value, the higher the rate of fat metabolism. Older visualization platforms do not allow researchers to see these kinds of changes in lipid metabolism within individual fat cells.
"Droplet size is a way to track how much of the stored fat is 'turning over' or getting converted back into energy. This is an important aspect of lipid metabolism, and we now have a tool that allows us to track changes in the size of specific lipid droplets within individual cells of fruit flies," said Yajuan Li, MD. PhD, who is a postdoctoral researcher in the Shi lab at UC San Diego and the first author on the paper in Aging Cell.
Heavy water
The new visualization platform builds on some of Shi's earlier work using a variation on regular water, called heavy water or (D2O). Heavy water is, literally, heavier than regular water. Heavy water molecules contain one oxygen atom like regular water. But in place of the pair of hydrogen atoms—the "H2" in "H20"—heavy water contains a pair of heavier deuterium atoms.
Like "regular" water, heavy water is freely incorporated into cells in living organisms. So when the researchers provide heavy water to a fruit fly, and then that fruit fly begins to convert energy from its food into fat molecules to be stored, some of those fat molecules contain deuterium. In this way, the prevalence of deuterium atoms in lipids stored within the fat cells of fruit flies provides a way to measure how much fat that fly has stored.
By changing a fly's diet at the same time that you introduce heavy water, you have a way to monitor how the diet changes lipid turnover. More details on how the system works are in this 2021 profile, in which Shi said, “When we are developing a new technology, a new tool, it will definitely inspire us to ask new biological questions.”
When it comes to understanding the connections between diet composition and lipid metabolism, the new biological questions are bringing researchers back to some of the oldest and most intriguing questions about links between diet and obesity, diabetes, aging and longevity.
Paper title: "DO-SRS imaging of diet regulated metabolic activities in Drosophila during aging processes" in the journal Aging Cell on March 7, 2022. Authors: Yajuan Li, Wenxu Zhang, Anthony A. Fung, and Lingyan Shi from the Department of Bioengineering at the University of California San Diego Jacobs School of Engineering. Funding: NIH, Grant/Award Number: U54 pilot grant 2U54CA132378; Jacobs School of Engineering, University of California San Diego; Hellman Fellow Award from UC San Diego.
Share This:
You May Also Like
Stay in the Know
Keep up with all the latest from UC San Diego. Subscribe to the newsletter today.


