
Unexpected Activity of Two Enzymes Helps Explain Why Liver Cancer Drugs Fail
Overturning previous assumptions, study also provides new, more realistic model for liver cancer research and drug development
Published Date
By:
- Heather Buschman
Share This:
Article Content
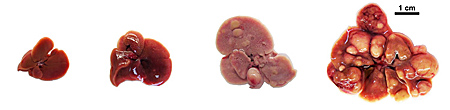
Left to right: 1) healthy mouse liver, 2) liver deficient for Shp2, 3) liver deficient for Pten, 4) liver deficient for both Shp2 and Pten. Tumors are visible in 3) and are even more prominent in 4), where both enzymes are lacking.
Some cancers are caused by loss of enzymes that should keep cell growth in check. On the flip side, some are caused by over-activation of enzymes that enhance cell growth. Yet drugs that inhibit the overactive enzymes have failed to work against liver cancer. In mouse models, researchers at University of California San Diego School of Medicine have discovered a potential reason — counterintuitively, lack of both types of these enzymes can lead to liver disease and cancer. In human liver tumor samples, they also found that deficiencies in these two enzymes, called Shp2 and Pten, are associated with poor prognosis.
The study, published December 13 by Cell Reports, provides a new understanding of how liver cancer develops, a new therapeutic approach and new mouse model for studying the disease.
“When it comes to liver cancer, I think we’ve been making strategic mistakes,” said senior author Gen-Sheng Feng, PhD, professor of pathology and biological sciences at UC San Diego. “In cancer development, we always thought about two distinct families of enzymes — one promotes cancer, one inhibits it. Many drugs have been developed to block the cancer-promoting pathways, but we and others are now finding that many classical pro-cancer proteins are actually inhibitors.”
Based on Shp2’s well-known role in promoting tumor formation, researchers have long assumed that drugs that block the enzyme would inhibit tumor formation. But Feng’s team previously found the opposite to be true — loss of the Shp2 enzyme can promote liver cancer. In contrast, in a study published last year, Feng and team discovered that removing both Shp2 and Pten, a tumor-suppressing enzyme, neutralized leukemia development.
“So the roles of tumor-promoting and tumor-suppressing enzymes are not as simple as we thought,” Feng said. “This also explains many unwanted side effects with drugs that target these enzymes. Their consequences can differ depending on cell type.”
In this latest study, Feng and team found that Shp2 and Pten cooperate to suppress liver tumor formation in experimental mice. When the researchers deleted both the Shp2 and Pten genes specifically in the mice’s liver cells, early-onset liver disease (non-alcoholic steatohepatitis, or NASH) was more severe and liver tumors occurred earlier and more frequently than in control mice with one or both enzymes functioning.
Normal mice did not experience any tumors. In mice lacking either Pten or Shp2, liver tumors began to appear after approximately seven or 12 months. But for mice lacking both enzymes, 80 percent spontaneously developed liver tumors in five months; 100 percent had tumors at seven months.
The increased severity of liver disease and frequency of liver cancer in these models is likely because lack of Shp2 and Pten enzymes activates molecules involved in lipid metabolism, inflammation and fibrosis, the researchers said.
After their surprising results in mice, the researchers wondered if the same phenomenon occurs in human liver cancers. They analyzed 335 human liver tumor samples and found that approximately 52 percent of the tumors were low in both Shp2 and Pten enzyme levels. Those patients with low Shp2 and Pten enzyme levels in the tumors had a poorer prognosis than those with higher levels of one or both enzymes — at 50 months after surgery, approximately 60 percent of patients with low Shp2 and Pten were alive, compared to approximately 90 percent of patients with high Shp2 and Pten levels.
These results provide new information about how liver cancer arises and a new target for drug development. But the findings also offer a new laboratory model for studying the disease. Previously, Feng’s team and others would use chemical carcinogens to induce liver cancer in mouse models. Instead of taking this artificial approach, Feng said researchers can now generate more realistic mouse models of non-alcoholic fatty liver disease and liver cancer simply by removing the Pten and Shp2 enzymes.
“Liver cancer is more complicated than we thought. These pathways, when over-activated, stimulate tumor development, but so does inhibiting them,” Feng said. “That’s why we can’t rush to conclusions like we have in the past. But now that we have a good model that mimics the human pathogenic process and we can use that to work out the mechanisms that lead to liver disease and cancer, and search for novel drug targets.”
Co-authors of this study include: Xiaolin Luo, Kaisa L. Hanley, Helen He Zhu, Kirsten N. Malo, Carolyn Hernandez, Nissi M. Varki, Nazilla Alderson, Catherine Chu, Shuangwei Li, Rohit Loomba, UC San Diego; Rui Liao, UC San Diego, Fudan University, and Chongqing Medical Unversity; Xufu Wei, UC San Diego, and Chongqing Medical University; Jia Fan, Shuang-Jian Qiu, Fudan University.
This research was funded, in part, by the National Institutes of Health (R01CA176012, R01CA188506) and UC San Diego-Roche Extending Innovation Network.
Share This:
You May Also Like
Stay in the Know
Keep up with all the latest from UC San Diego. Subscribe to the newsletter today.


