Mini-brain Model of Idiopathic Autism Reveals Underlying Pathology of Neuronal Overgrowth
Published Date
By:
- Scott LaFee
Share This:
Article Content
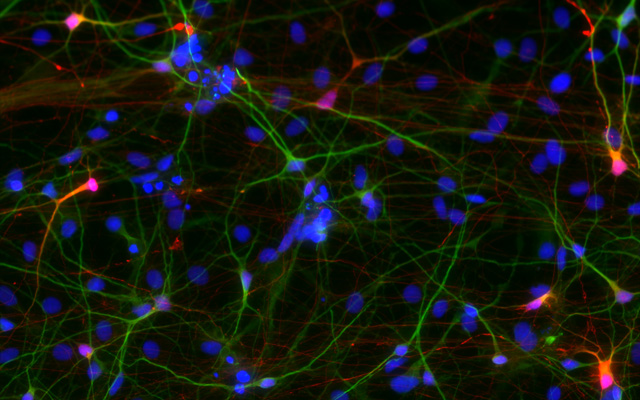
The vast majority of cases of autism spectrum disorder (ASD) are idiopathic – the cause is unknown. In a paper published this month in the journal Molecular Psychiatry, researchers at the University of California San Diego School of Medicine, with colleagues across the nation and world, have created a “mini-brain” model, derived from persons with a particular form of idiopathic ASD characterized by over-sized brains, revealing a defective molecular pathway during brain development that results in early neuronal overgrowth and dysfunctional cortical networks.
“The bottom line is that we can now effectively model idiopathic ASD using a cohort of individuals selected by a clear endophenotype. In this case, brain volume,” said senior author Alysson R. Muotri, PhD, associate professor in the UC San Diego School of Medicine departments of Pediatrics and Cellular and Molecular Medicine. “And early developmental brain enlargement can be explained by underlying molecular and cellular pathway dysregulation, leading to altered neuronal cortical networks.”
The characteristics and causes of ASD are diverse and not entirely known – facts that have made it difficult to fully uncover relevant genetic, pathologic and cellular factors that might be broadly shared. One distinct pathophysiology or disordered process is the occurrence of macrencephaly in some children with ASD, which is characterized by early neuronal overgrowth and abnormally enlarged brains. Macrencephaly occurs in the first three years of life and precedes the first clinical signs of ASD. Approximately 20 percent of ASD individuals have macrencephaly.
The researchers reasoned that ASD individuals with macrencephaly likely shared an underlying molecular and cellular pathology. They created neural progenitor cells programmed from induced pluripotent stem cells derived from children with ASD.
“By genome sequencing, we realized that some, but not all, carried clear mutations in the Wnt pathway, which is a molecular pathway previously implicated in cancer,” said Muotri. “Defects on cell cycle control were also obvious from gene expression on these cells. As a consequence, neural progenitor cells derived from these kids proliferate faster than controls, explaining the big brain phenotype.”
Next, the researchers differentiated the progenitor cells into networks of cortical neurons, the primary functional cell type of the brain’s cortex (gray matter).
“We showed that ASD networks fail to produce inhibitory neurons and found that several receptors and neurotransmitters related to GABA (an amino acid that acts as a neurotransmitter) are misregulated on these neurons. We also showed that the number of excitatory synapses is reduced, leading to functional defects when we analyzed the maturation of neuronal networks over time. Basically, we detected a lack of burst synchronization (when multiple neurons fire simultaneously).
Finally, the research team tested a drug already in clinical trials (IGF-1) on a cohort of study participants, finding that it provoked a reversal of neural alterations, though the degree of response varied by ASD individual.
Muotri said the findings show it is possible to more effectively stratify ASD individuals for clinical trials by identifying persons who are likely to be more responsive to specific therapies using their “mini-brains” in a dish.
Co-authors include: M.C. Marchetto, K.C. Vadodaria, A.P.D. Mendes, H. Ghosh, R. Wright and F.H. Gage, Salk Institute for Biological Studies; H. Belinson, UC San Francisco; Y. Tian, J. Ou and D.H. Geschwind UCLA; B.C. Freitas and C.A. Trujillo, UC San Diego and Rady Children’s Hospital-San Diego; C. Fu, Case Western Reserve University; P.C. Beltrao-Braga, UC San Diego, Rady Children’s Hospital-San Diego and University of Sao Paolo, Brazil; K. Padnanabhan, University of Rochester School of Medicine and Dentistry, NY; Y. Nunez, Salk Institute, UC San Diego and Rady Children’s Hospital-San Diego; K.J. Brennand, Icahn School of Medicine at Mount Sinai, NY; K. Pierce, L. Eichenfield, T. Pramparo, L.T. Eyler, C.C. Barnes and E. Courchesne, UC San Diego; and A. Wynshaw-Boris, UC San Francisco and Case Western Reserve University.
Funding for this research came, in part, from the California Institute for Regenerative Medicine, the National Institutes of Health (1-DP2-OD006495-01, R01 MH100175-01, U19MH107367) the International Rett Syndrome Foundation, NARSAD, Helmsley Trust, JPB Foundation, Engmann Foundation, CDMRP Autism Research Program and Autism Speaks.
Share This:
Stay in the Know
Keep up with all the latest from UC San Diego. Subscribe to the newsletter today.