
By:
- Mario Aguilera
Published Date
By:
- Mario Aguilera
Share This:
UC San Diego Scientists to Explore New Frontiers in Parkinson’s Disease Research with $7.2M Grant
Aligning Science Across Parkinson’s initiative supports international project aiming to unravel the biology of a gene linked to Parkinson’s disease
In August, a team of researchers at the University of California San Diego published groundbreaking back-to-back studies describing unprecedented details of a protein linked to genetically inherited Parkinson’s disease. The researchers produced the first visualizations of leucine-rich repeat kinase 2, or LRRK2, as seen within its natural environment inside the cell, as well as the first high-resolution blueprint of the protein.

Samara Reck-Peterson, lead principal investigator of the project.
The Aligning Science Across Parkinson’s (ASAP) initiative has announced support for the next phase of the research as the scientists focus on understanding the basic mechanisms underlying Parkinson’s, a neurodegenerative disorder affecting millions. ASAP’s goal is to support research that will inform a path to a cure for Parkinson’s. The three-year, $7.2 million grant will fund research across three UC San Diego laboratories and two others based in Germany. The Michael J. Fox Foundation for Parkinson’s Research is the implementation partner for ASAP and issuer of the grant, which contributes to the Campaign for UC San Diego.
“This grant from ASAP will further advance UC San Diego’s efforts at unraveling the core debilitating effects of Parkinson’s disease, which impacts the lives of so many individuals and families around the world,” said UC San Diego Chancellor Pradeep K. Khosla. “This support will keep our researchers at the forefront of the science and technology needed to fully understand the mechanisms underlying the disease.”
Since LRRK2 was discovered and linked to Parkinson’s in the early 2000s, scientists have vigorously pursued clues about its form and function. The new funding expands efforts at UC San Diego using leading-edge cryo-electron microscopy (cryo-EM) to produce previously unseen views of biologically important cells and molecules.
“The goal of this project is to understand the basic cell biology and structure of this really fundamentally important LRRK2 molecule,” said Samara Reck-Peterson, the lead principal investigator of the project and professor at UC San Diego School of Medicine and Division of Biological Sciences and a Howard Hughes Medical Institute investigator. “If we can find out why LRRK2—when it doesn’t work—causes Parkinson’s disease, that’s really the ultimate goal. When you are thinking about designing a drug, you really need to understand all the details of the parts in order to engineer therapeutics.”

Project co-principal investigator Andres Leschziner.
Project co-principal investigator Andres Leschziner and his colleagues have used the growing cryo-EM facility at UC San Diego to produce atomic-level visualizations of LRRK2 in the most detailed images of the protein to date. Leschziner plans to use cryo-EM to develop a full blueprint of LRRK2 in normal and mutant states.
“LRRK2 is a complicated molecule with a lot of moving parts, and its dynamic behavior is very likely to play a role in both its normal function and Parkinson’s pathology. Understanding how the structure of LRRK2 changes in different states and with different disease mutations will be key to developing treatments. The equipment and expertise in cryo-EM here at UC San Diego put us in a great position to visualize all of this,” said Leschziner, a professor at UC San Diego School of Medicine and Division of Biological Sciences.
Biological Sciences Assistant Professor and project co-principal investigator Elizabeth Villa uses a related technology called cryo-electron tomography (cryo-ET) to visualize LRRK2 in its natural living environment within the cell. In combination with other techniques, Villa’s lab will continue to examine mutant forms of LRRK2 as it interacts with a network of components in the cell in health and disease.
“We are just starting to understand the roles of LRRK2 in various cellular processes,” said Villa. “Using high-end technologies, we are, for the first time, able to directly see LRRK2 as it performs these roles, as well as what happens when mutations affect LRRK2’s function. By opening windows into LRRK2 in cells, we can answer longstanding questions and generate new ones. It is humbling and empowering to know that our basic research can benefit people who suffer from this debilitating disease.”

Project co-principal investigator Elizabeth Villa.
Reck-Peterson’s expertise focuses on “roadways” of tracks called microtubules that move important cargoes around the cell. Previous evidence suggests that LRRK2 plays a role in how these components move along these cellular tracks. Her lab will be investigating cargo movements when LRRK2 is normal and in diseased states, and whether interactions with microtubules are linked to Parkinson’s.
The LRRK2 project and the new funding are the latest achievements underscoring the university’s rising cryo-EM facility. Cryo-EM, in which scientists freeze molecules in a thin layer of ice to determine their structure at high resolution, has exploded in scientific prominence over the last decade as the technology provides unique insights into a range of biological phenomena.
“This project will build upon the university’s investments in cryo-EM technology and deliver new insights into Parkinson’s disease that promise to lead to new treatments,” said Division of Biological Sciences Dean Kit Pogliano. “I’m grateful to ASAP for recognizing that this all-star team of scientists is well-equipped to make transformational discoveries that will provide new insights into the biology of this devastating disease.”
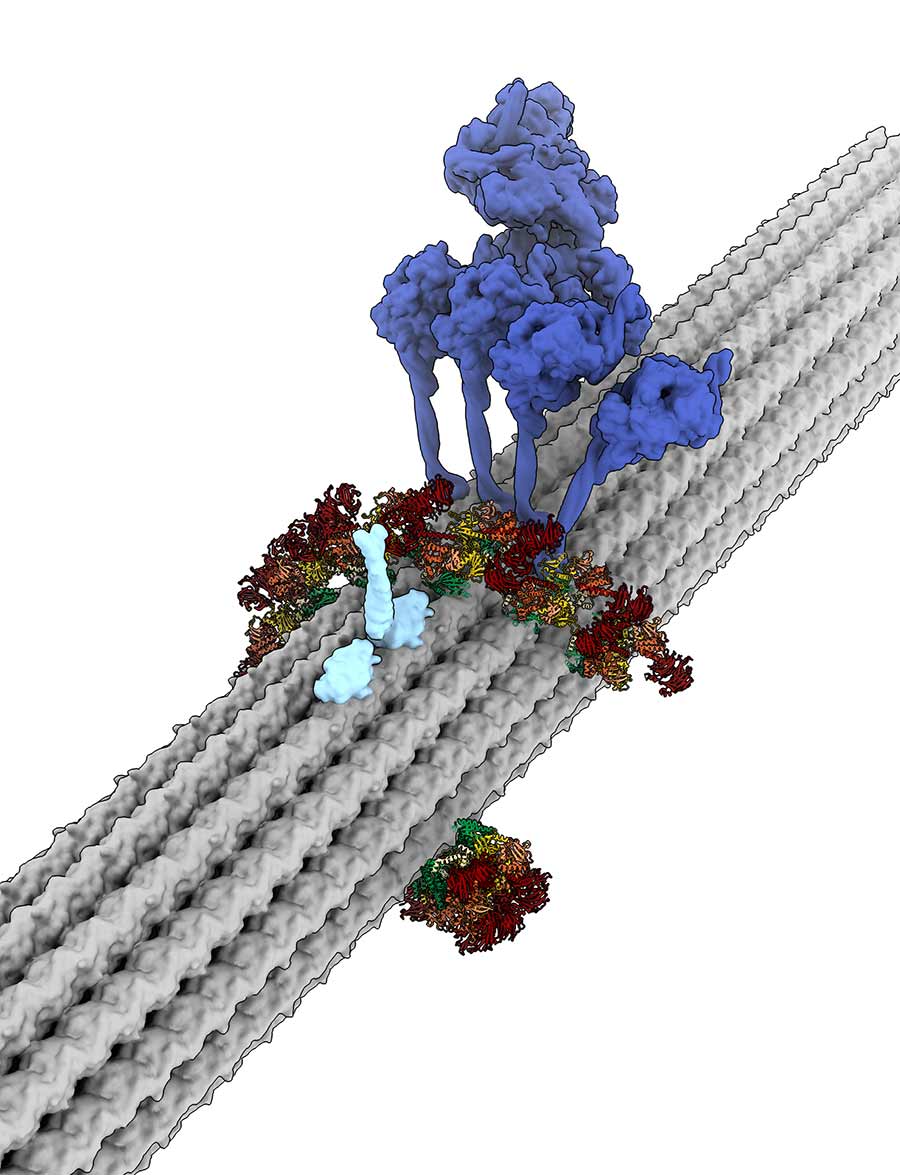
Roadblock: This image depicts a model for how LRRK2 could block critical movement across the cell. The visualization shows a short LRRK2 filament (red, green and yellow) wrapped around a cellular track called a microtubule, forming a roadblock for the microtubule-based motors kinesin (light blue) and dynein (blue).
Co-principal investigator Stefan Knapp, a professor at Goethe University in Frankfurt, Germany, will be engineering samples of LRRK2 that the team members can use to help decode its full structure, and will also design probes to locate LRRK2 inside cells for both live-cell imaging and cryo-ET. The fifth team member, Florian Stengal of Konstanz University in Germany, brings expertise in mass spectrometry, an analytical tool that will help the team develop a complete picture of all of the proteins LRRK2 interacts with in normal and Parkinson’s cells.
“All five team members are going to be working in their specialties but toward our common goals, and there’s going to be a lot of crosstalk among the team,” said Reck-Peterson. “One of the things that we’re really excited about is that every member of the team brings a unique strength, and we’ve already shown that we are really good at working together given our track record of collaboration.”
The new funding allows the UC San Diego labs to rapidly expand their teams focusing on LRRK2 research. Initial phases of this research were funded by The Michael J. Fox Foundation for Parkinson’s Research in an effort spearheaded by UC San Diego’s Susan Taylor, a distinguished professor in Chemistry and Biochemistry and Pharmacology and world-renowned expert in protein kinases, one of the largest gene families to which LRRK2 belongs.
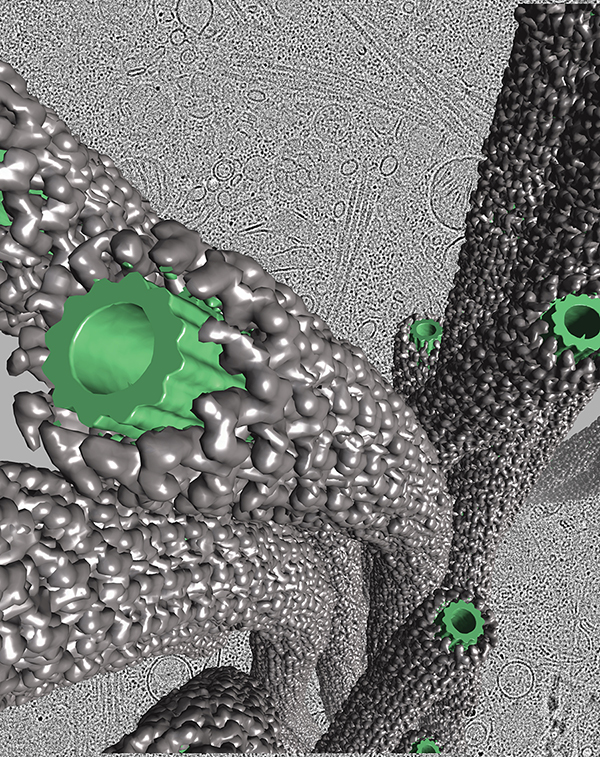
The molecular structure of a Parkinson’s disease mutant LRRK2 was captured inside cells using advanced cryo-electron tomography and computational modeling techniques. Here, a window into a human cell carrying an LRRK2 mutation is shown in grayscale in the background. By zooming in, the structure of this important drug target was revealed (gray) decorating molecular highways called microtubules (green).
“UC San Diego has a long and accomplished history in uncovering fundamental secrets about how key proteins function in health and diseases—kinases in particular,” said Dr. David A. Brenner, Vice Chancellor of Health Sciences at UC San Diego. “In more recent years, we’ve made it a strategic goal to take those efforts to the next level by first recruiting the nation’s rising stars in protein structure and function and then providing them access to leading-edge technologies such as cryo-EM. I am thrilled to see the success of this reflected in the incredible work of this team.”
Private support, like the grant from Aligning Science Across Parkinson’s (ASAP) initiative, contributes to the Campaign for UC San Diego—a university-wide comprehensive fundraising effort concluding in 2022. Alongside UC San Diego’s philanthropic partners, the university is continuing its nontraditional path toward revolutionary ideas, unexpected answers, lifesaving discoveries and planet-changing impact. Learn more at Campaign for UC San Diego.
Share This:
You May Also Like
Stay in the Know
Keep up with all the latest from UC San Diego. Subscribe to the newsletter today.


