
Biologists Use Flies and Mice to Get to the Heart of Down Syndrome
By:
- Kim McDonald
Published Date
By:
- Kim McDonald
Share This:
Article Content
A novel study involving fruit flies and mice has allowed biologists to identify two critical genes responsible for congenital heart defects in individuals with Down syndrome, a major cause of infant mortality and death in people born with this genetic disorder.
In a paper published in the November 3 issue of the open access journal PLoS Genetics, researchers from UC San Diego, the Sanford-Burnham Medical Research Institute in La Jolla, Calif., and the University of Utah report the identification of two genes that, when produced at elevated levels, work together to disrupt cardiac development and function.
Down syndrome, the most common genetic cause of cognitive impairment, is a disorder that occurs in one in 700 births when individuals have three, instead of the usual two, copies of human chromosome 21.
“Chromosome 21 is the shortest human chromosome and intensive genetic mapping studies in people with Down syndrome have identified a small region of this chromosome that plays a critical role in causing congenital heart defects,” said Ethan Bier, a biology professor at UC San Diego and one of the principal authors of the study. “This Down syndrome region for congenital heart disease, called the ‘DS-CHD critical region,’ contains several genes that are active in the heart which our collaborator, Julie Korenberg, had suspected of interacting with each other to disrupt cardiac development or function when present in three copies. But exactly which of these half dozen or so genes are the culprits?”
“Identifying the genes within the DS-CHD critical region contributing to congenital heart defects is challenging to address using traditional mammalian experimental models, such as mice,” added Bier, “since the number of possible genetic combinations that would need to be generated and tested is very large.”
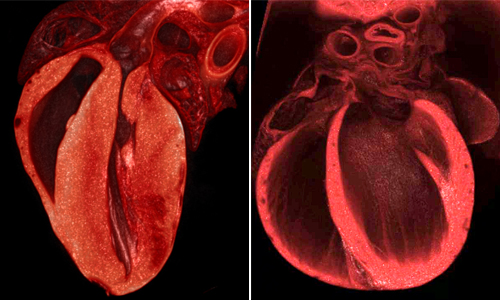
Elevated levels of the two genes produce a thickening of the ventricle walls in a mouse heart at left. At right, a normal mouse heart. Credit: UC San Diego
To simplify their search, the scientists turned to fruit flies, a simpler and rapidly reproducing biological system with many of the same genes as mice and humans. With help from collaborators Amir Gamliel, Geoff Rosenfeld and Kirk Peterson at the UC San Diego School of Medicine, Rolf Bodmer and Karen Ocorr at the Sanford-Burnham Medical Research Institute, and Julie R. Korenberg at the University of Utah, biologist Tamar Grossman in Bier’s lab devised a sequential genetic approach to untangle the problem.
“First, fruit flies were used to test for all possible pairwise genetic interactions between these genes that might disrupt the function of the simple fluid pumping fly heart,” said Bier.” These comprehensive genetic studies pointed to a particular pair of genes known as DSCAM and COL6A2 that resulted in the most severe defects when over-produced together.”
Then the researchers tested the effects of increasing the levels of these genes in the hearts of experimental mice. They first generated genetic lines of mice having elevated activity of each of these genes in the heart and then genetically crossed these mice to create offspring that over-produced both genes together. The parental mice as well as their offspring were then tested for heart function and visible heart defects.
Mice having elevated levels of each gene separately were largely normal. But the offspring with extra levels of both genes suffered from severe cardiac defects. These heart defects were of two kinds. The first resembled one of the salient features of Down syndrome cardiac patients, in which blood shunts between the two atrial chambers of the heart through small holes in a septum that normally isolates these two chambers. The second defect, which is not frequently observed in Down syndrome patients, but is a common and very serious condition in the general population, was a thickening of the heart wall—referred to medically as cardiac hypertrophy.
“Such thickening of the heart wall greatly reduces heart function and can lead to fatal heart attacks, which indeed was observed among some of the more seriously affected DSCAM and COL6A2 over-producing mice,” said Bier.
Bier added that the tiered genetic approach, using fruit flies, then mice, could be useful in identifying genes involved in other common genetic disorders that are thought to be caused by multiple genes.
“These conditions arise due to a surprising variation in the copy number of small intervals of human chromosomes that are carried by virtually all people,” he said. “Depending on which small regions of the chromosome have extra or fewer copies of genes, various conditions can result including obesity, autism, and schizophrenia. Typically in these diseases, as in Down syndrome, the difficult puzzle is which of the possible genes with altered copy number are involved in causing the disease.”
Funding for the research project was provided by grants from the National Institutes of Health.
Share This:
You May Also Like
Stay in the Know
Keep up with all the latest from UC San Diego. Subscribe to the newsletter today.


