
Now Showing: Medicinal Chemistry in 3D
Published Date
By:
- Cynthia Dillon
Share This:
Article Content
In the movie industry, filmmakers often turn to books for stories to entertain audiences. In the chemistry biz, drug discoverers draw from libraries—fragment libraries—to produce new drugs for treating illnesses like the flu or cancer. These collections of small portions of molecules offer researchers a decent amount of chemical diversity to work with, but they don’t offer a lot of variety in shape—mostly one- and two-dimensional fragments.
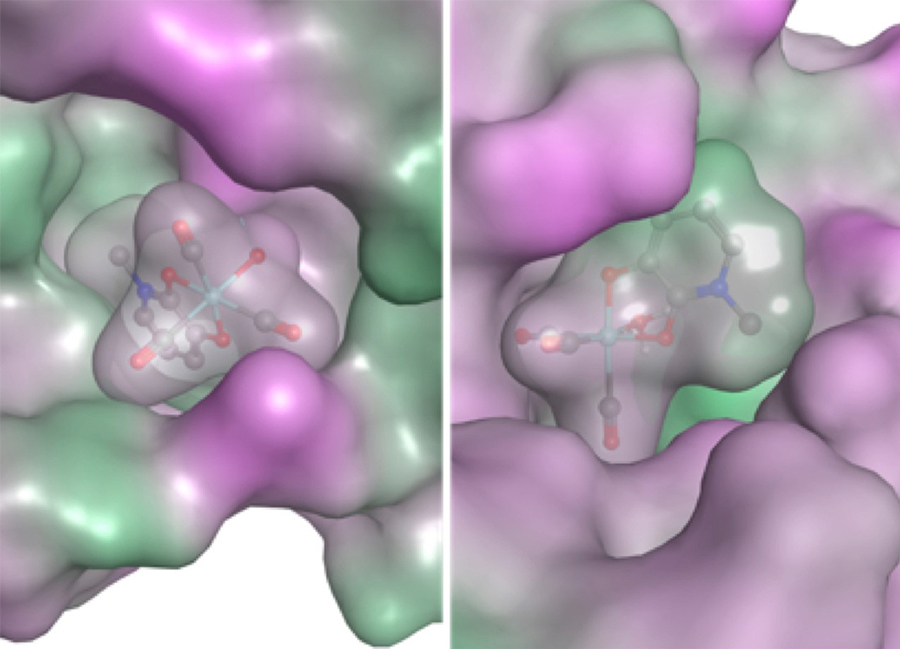
In the image, fragments and protein are shown with molecular surface maps colored to indicate lipophilicity. Hydrophilic and lipophilic regions are represented by pink and green surfaces, respectively. Figure courtesy of Seth Cohen, UC San Diego
Yet, according to research by UC San Diego’s Seth Cohen published in Chemical Science and highlighted in Chemistry World, shape plays a key role in dictating the biological effects of molecules—namely recognition by a biological target. So he and his research team, including colleagues at Germany’s Ruhr University Bochum led by Nils Metzler-Nolte, used a new type of metal-based molecular building block to serve as the basis for developing new therapeutic drugs.
“The reason we used these is because they have unique shapes that are not readily achieved by organic, or metal-free, molecular building blocks. These unique shapes allow them to bind to important biomolecules for treating disease,” explained Cohen. “This research could open up new avenues for discovering drugs to treat life-threatening illnesses such as cancer, influenza or multidrug resistant bacteria.”
Comparing these molecular building blocks to keys that need to fit in a keyhole, the professor of chemistry and biochemistry noted that most keys—organics in chemistry—are flat and only will fit in “conventional” locks. “Our new metal-based building blocks are like the circular, three-dimensional (3D) keys found on some bike or computer locks. Therefore, using our ‘new keys’ we can access locks that conventional keys cannot fit into,” he said.
While science’s bioinorganic community has explored the use of coordination and organometallic compounds before, it has not done so in the context of fragment-based drug discovery (FBDD), which is a powerful strategy for the identification of new bioactive molecules. Combining their passion and expertise in inorganic and medicinal chemistry in a new and creative way, Cohen’s team focused on 3D mononuclear complexes as structural scaffolds that can be grown into more drug-like molecules by modifying specific parts of the fragments. The work yielded a novel metallofragment (mF) library featuring 13 classes of complexes. These were screened against three therapeutic targets: portions of the H1N1 influenza A virus, NDM-1 (found in some antibiotic-resistant bacteria), as well as an anticancer target. The screenings validated the suitability of the mF platform for future FBDD efforts.
“We hope, in an ideal scenario, that in eight to 10 years’ time, some of these building blocks will begin to find their way into new drug candidates that are providing life-saving cures for human health,” said Cohen.
This research was supported by the National Institutes of Health (R21 AI138934 and F32 GM125233), the University of California President’s Postdoctoral Fellowship, a Natural Sciences and Engineering Research Council of Canada Postdoctoral Fellowship and the National Science Foundation Graduate Research Fellowship Program (DGE-1650112).
Share This:
You May Also Like
Stay in the Know
Keep up with all the latest from UC San Diego. Subscribe to the newsletter today.

