
UC San Diego/SDSC Study Advances Brain Cancer Research
Findings Pinpoint Modules that Regulate Glioblastoma Genes
Published Date
By:
- Lynne Friedmann
Share This:
Article Content
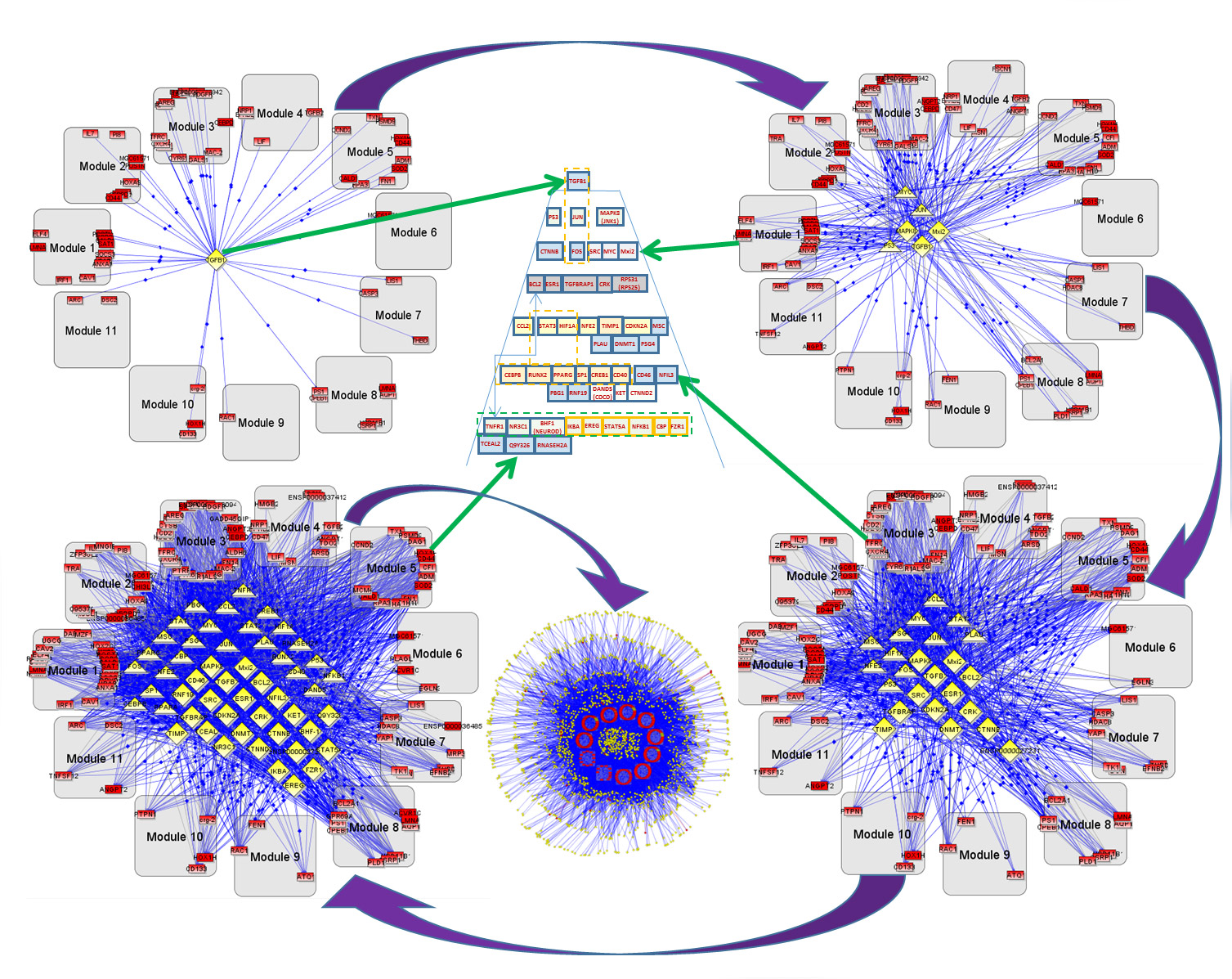
This image shows transcription regulators on the different levels of hierarchy that control the specified coherent gene modules responsible for glioblastoma. Valentina L. Kouznetsova, SDSC.
Researchers at the San Diego Supercomputer Center (SDSC), Moores Cancer Center, and Department of Neurosciences at the University of California, San Diego, have shown for the first time a pyramid hierarchical network of “coherent gene modules” that regulate glioblastoma genes, involved in a highly aggressive form of brain cancer.
By identifying the most important gene modules responsible for cancer growth and proliferation, the study informs a strategy that could elucidate these modules at the top levels of their network, and in turn be used to identify new drug therapies. The paper appears in the U.K. journal Molecular BioSystems, published by the Royal Society of Chemistry.
Glioblastoma (GBM) is highly resistance to treatment and rapidly lethal. To date, therapeutic targeting of individual molecules has proven unsuccessful in treating GBM due to complex interactions of genetic pathways, signaling networks, and cellular levels that are a hallmark of the cancer and allow it to proliferate.
“Considering the complex interplay of mediators involved, it is clear that examining individual signaling molecules or even pathways may not give information accurately representing the underlying molecular abnormalities,” said lead author Igor F. Tsigelny, a research scientist with SDSC as well as the UC San Diego Moores Cancer Center and the Department of Neurosciences.
Because of the number of connections involved, researchers relied on the BiologicalNetworks server that used SDSC’s data-intensive Gordon supercomputer. The test runs of this server queries on Gordon showed up to a 200x application speed improvement. Of particular interest in the study was elucidation of transcription regulators (TRs) and transcription factors (TFs) that actually regulate the coherent gene groups and the drug targeting that can be applied to these controlling TRs and TFs.
“Our approach of identifying major multi-level hierarchical networks and coherent gene modules within networks improves our understanding for clinical translation,” said Tsigelny.
“This innovative network analysis identified several central nodes in signaling networks in glioblastoma that could help us design personalized clinical trials using available and new drugs in development,” said Santosh Kesari, Director of Neuro-Oncology and a professor of neurosciences at the Moores Cancer Center.
In addition to SDSC’s Tsigelny, researchers involved in the study include Valentina L. Kouznetsova from SDSC; and Penfgei Jiang, and Sandeep C. Pingle from the Moores Cancer
Share This:
You May Also Like
Stay in the Know
Keep up with all the latest from UC San Diego. Subscribe to the newsletter today.


