
Wired to Think
UC San Diego leads study to advance knowledge of brain signaling with breakthrough sensors
Published Date
By:
- Cynthia Dillon
Share This:
Article Content
To understand how humans think, it comes down to the wiring. For 60 years, scientists have used metal electrodes—recording sensors—to study cognition by capturing and measuring electrical spikes of a few neurons during brain activity. This gives them a partial picture of brain signals. To get a full picture of thought processes at work, however, UC San Diego Neurophysicist David Kleinfeld asserts that new electrode sensors that make use of exotic structures are needed.
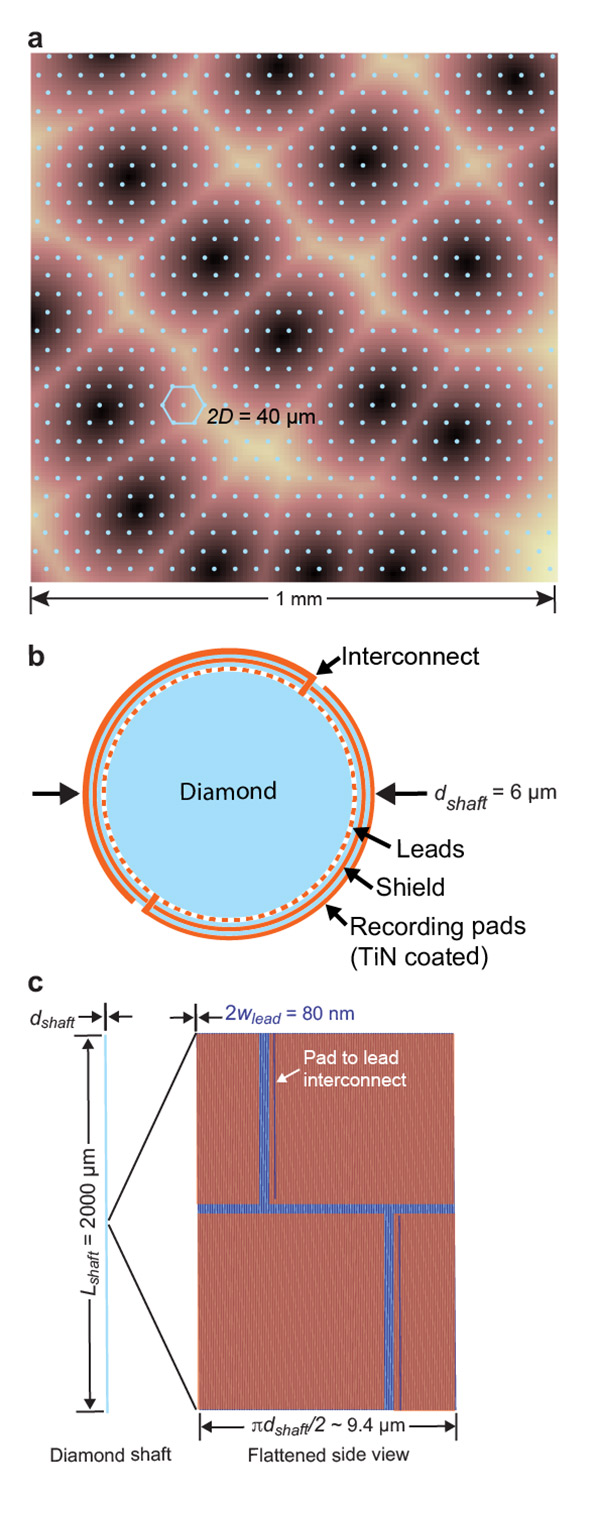
Figure 1. Schematic of the Proposed Stiff Probe. (A) A grid of the proposed electrodes superimposed on top of mouse cortex; the dark regions correspond to regions that process input from individual whiskers from the mouse. (B) The electrode is constructed of a diamond shaft that is covered with five layers; electrode leads; mineral insulation; a conducting shield; mineral insulation; electrodes. (C) The multi-electrode shaft with an expanded view of the face to show the arrangement of the electrode pads. Figure courtesy of David Kleinfeld, UC San Diego
To prove the point, Kleinfeld and a team of researchers analyzed the anatomical, electrical, mechanical, physiological, signaling and thermal constraints that need to be considered for the case of measuring electrical spikes from all neurons in the cortex of model mammalian brains, using microwire electrodes. They concluded that it is possible to fabricate rigid shanks with multiple electrodes that record electrical signals from every neuron in the cortex—at the same time. Their study, published recently online in Neuron, supplies a blueprint for a future generation of electrode sensors—notably microscopically slender diamond needles—that utilizes existing yet nontraditional materials and fabrication procedures.
“It simply blows your mind to realize that atoms and molecules have coalesced to form living matter that not only reproduces, but seeks to understand its own properties,” said Kleinfeld. “We need to understand the nature of this intelligence. Technological innovation is a key component of forward progress.”
With inspiration from two workshops—a 2014 National Science Foundation (NSF)-funded workshop called, “Limits of Measurements from Brains,” co-organized by Partha Mitra of Cold Spring Harbor Laboratory, and a subsequent workshop at the Howard Hughes Medical Institute (HHMI) Janelia Research Campus in 2017, co-organized by Kleinfeld, Mitra and Professor Timothy Harris of HHMI—Kleinfeld and a team of senior scientists and technologists asked the question: Can one concurrently record electrical spikes from every neuron in the brain?
“Far from a philosophical issue, the answer to this question stands as an essential guide toward deciphering the nature of signal processing between brain cells and estimating the computational power of the mammalian brain,” said Kleinfeld.
According to Mitra, a Harvard-trained theoretical physicist, brains pack a very large number of electrical signal-carrying “wires” (i.e., axons) together into a small space; so the approach proposed does not violate any physics principles, and it is a worthwhile engineering challenge.
Kleinfeld, a professor with appointments in the Department of Physics and the Section of Neurobiology at UC San Diego, said that after physically unfeasible approaches like embedding a dense array of nanoradios were set aside, attention was largely focused on optical imaging techniques.
“However, the natural light scattering properties of live brains gives rise to fundamental physics limits that constrain such an approach,” said Kleinfeld.
So the collaborators conducted a feasibility study that re-examined a classical approach—recordings from wire microelectrodes. They concluded that such an approach is feasible for recording from all, or a significant majority of, neurons in the cortices of rodents and small primates. The collaborators also noted the additional need to advance materials for improved flexible electrodes as an essential breakthrough for recording from the neurons in the brainstem and spinal cord in moving animals.
Other researchers who participated in the study included Kenneth Shepard of Columbia University, Rahul Sarpeshkar of Dartmouth, Lau Luan and Chong Xie of UT Austin, and Jacob Robinson of Rice University.
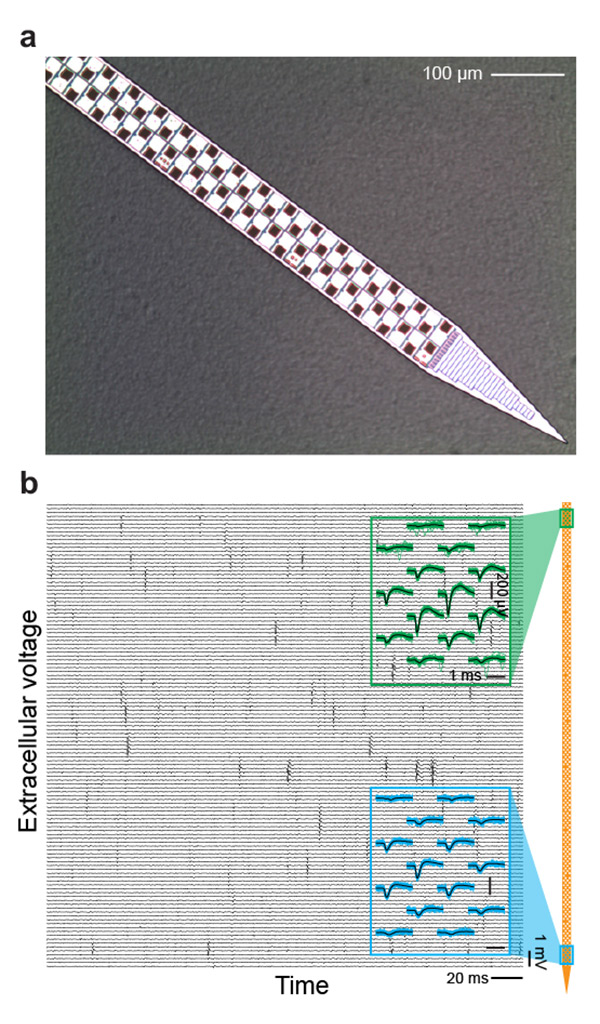
Figure 2. The Current State-of-the-Art Rigid Probe. (A) Photograph of the distal 68 sites of the 960 electrodes on a shank of a Neuropixels probe. The shank is 10 mm long, 70 µm wide, and 24 µm thick, with 12 µm by 12 µm titanium nitridecoated recording sites pitched at 2 per 20 µm of shank length. (B) Two example recordings from Neuropixels probe pads implanted in mammalian model cortex. Blue traces and green traces are 30 raw traces in the vicinity of a spike near the top (green) and bottom (blue) of the probe; black lines are average of those traces. Figure courtesy of David Kleinfeld, UC San Diego
“I work in an institute replete with classically trained biologists and physicists,” said Harris. “It is simply this great and raucous mixture of scientific cultures that can drive ideas and technology forward.”
According to Kleinfeld, the challenge of simultaneously recording the output signal, or electrical spike, from every neuron was one that partly inspired the The Brain Initiative, which funds basic research in neuroscience through the National Institutes of Health (NIH) and the NSF.
This work was supported by the Cold Spring Harbor Laboratory Crick-Clay Professorship, the Dr. George Feher Experimental Biophysics Endowed Chair, the Howard Hughes Medical Institute; the Neuroscience Eye Institute (grant EY028397); the National Heart, Lung and Blood Institute (grant HL140153); the National Institute of Neurological Disorders and Stroke (grants NS107466, NS109361 and NS0905905) and the Office of Naval Research (grant N00014-19-1-24340).
Share This:
You May Also Like
Stay in the Know
Keep up with all the latest from UC San Diego. Subscribe to the newsletter today.


